The paper was a collaborative group effort with several groups in Mainz, to which Denise contributed great work. Congratulations!
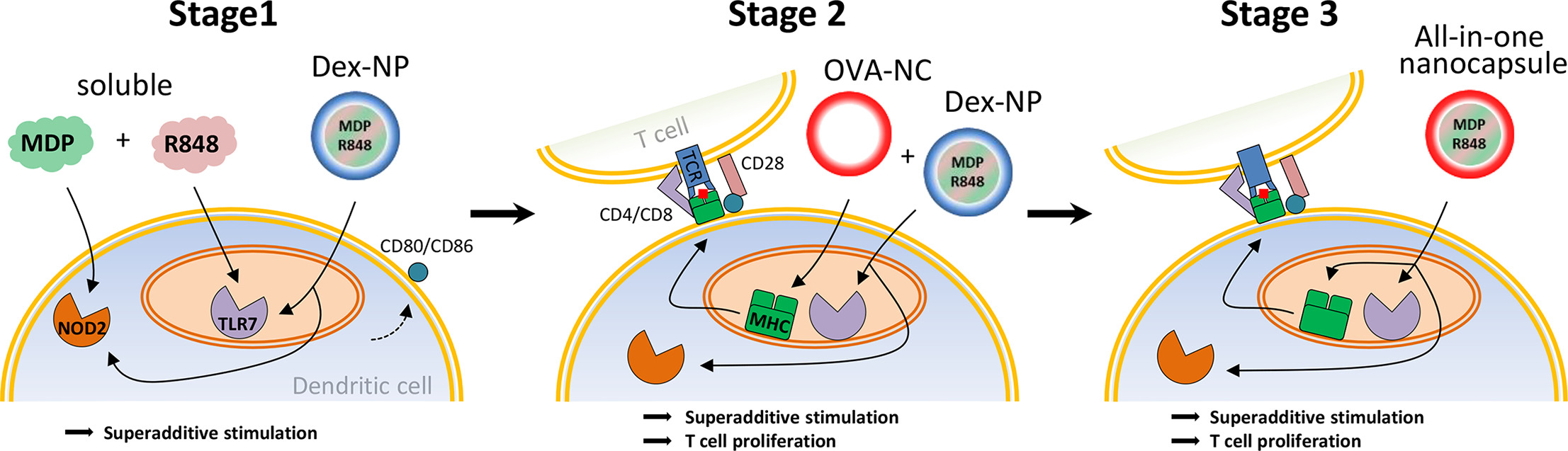
The paper describes the stepwise development of a nanocapsule for vaccination approaches, comprising a shell consisting of antigen and loaded with a superadditive adjuvant combination.
“Delivering all in one: Antigen-nanocapsule loaded with dual adjuvant yields superadditive effects by DC-directed T cell stimulation”
D. Paßlick, K. Piradashvili, D. Bamberger, M. Li, S. Jiang, D. Strand, P. R. Wich, K. Landfester, S. Grabbe, M. Bros, V. Mailänder
J. Controlled Release. 2018, just accepted (DOI: 10.1016/j.jconrel.2018.09.008)
Abstract:
Therapeutic vaccination is and remains a major challenge, particularly in cancer treatment. In this process, the effective activation of dendritic cells by a combination of distinctly acting adjuvants and an antigen is crucial for success. While most common vaccine formulations lack the efficiency to trigger sufficient T cell responses in a therapeutic tumor treatment, nanovaccines offer unique properties to tackle that challenge.
Here, we report the stepwise development of a nanocapsule for vaccination approaches, comprising a shell consisting of antigen and loaded with a superadditive adjuvant combination. In a first initial step, we identified the combination of resiquimod (R848) and muramyl dipeptide (MDP) to have a superadditive stimulatory potential. Particulated in Spermine-modified dextran-nanoparticles, the dual-adjuvant maintains its superadditive character and stimulates murine dendritic cells (DC) stronger than the soluble equivalents. The second step was to evaluate a protein-based nanocapsule as suitable antigen source for the induction of antigen-specific T cell responses. Therefore, the DC-mediated antigen-specific T cell proliferation upon treatment with nanocapsules, whose shell consists of ovalbumin (OVA), was assessed. At least, the superadditive adjuvant combination was encapsulated into OVA-nanocapsules to create the final nanovaccine. Its immunostimulatory potential for DC was extensively tested by measuring the expression of co-stimulatory surface markers, the secretion of pro-inflammatory cytokines and the capability to mediate OVA-specific T cell responses.
The developed nanovaccine triggers strong superadditive dendritic cell stimulation and potent antigen-specific CD4+ and CD8+ T cell proliferation. Combined with a high modifiability, an excellent biocompatibility, low cytotoxicity and an enormous loading capacity, the introduced antigen-nanocapsule provides an enormous potential for the effective delivery of superadditive adjuvant combinations, particularly when they target intracellular receptors.