“Development of Novel Peptide-based Michael Acceptors Targeting Rhodesain and Falcipain-2 for the Treatment of Neglected Tropical Diseases (NTDs)”
S. Previti, R. Ettari, S. Cosconati, G. Amendola, K. Chouchene, A. Wagner, U. A. Hellmich, K. Ulrich, R. L. Krauth-Siegel, I. Schmid, P. R. Wich, T. Schirmeister, J. Gut, P. J. Rosenthal, S. Grasso, M. Zappalà
J. Med. Chem. 2017, just accepted. (DOI: 10.1021/acs.jmedchem.7b00405)
Abstract:
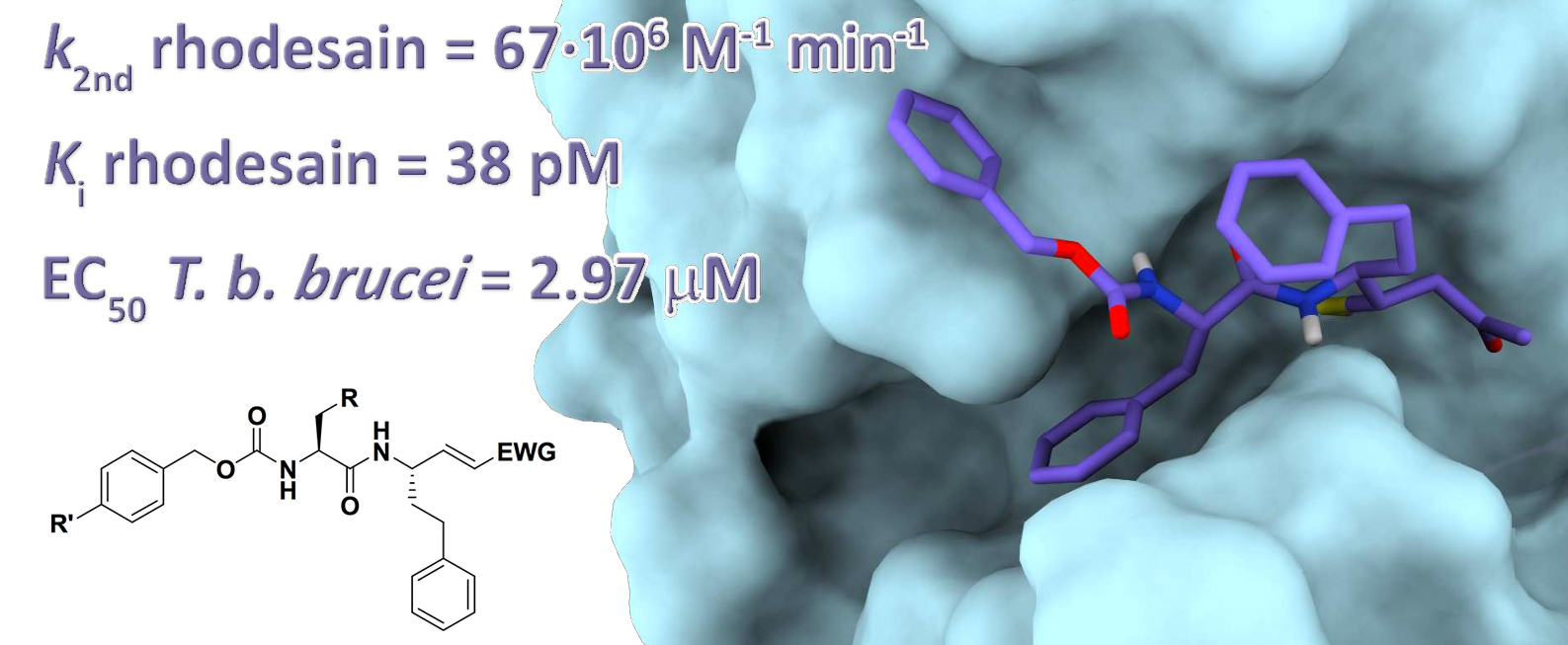
This paper describes the development of a class of peptide-based inhibitors as novel antitrypanosomal and antimalarial agents. The inhibitors are based on a characteristic peptide-sequence for the inhibition of the cysteine proteases rhodesain of T. b. rhodesiense and falcipain-2 of P. falciparum. We exploited the reactivity of novel unsaturated electrophilic functions such as vinyl-sulfones, -ketones, -esters and –nitriles. The Michael acceptors inhibited both rhodesain and falcipain-2, at nanomolar and micromolar level, respectively. In particular, the vinyl ketone 3b has emerged as a potent rhodesain inhibitor (k2nd= 67•106 M-1 min-1), endowed with a picomolar binding affinity (Ki = 38 pM), coupled with a single-digit micromolar activity against T. b. brucei(EC50 = 2.97 µM), thus being considered as a novel lead compound for the discovery of novel effective antitrypanosomal agents.